Gummy bears, those delightful, chewy little treats, have been a staple in candy jars and on store shelves for decades. With their vibrant colors and fun shapes, they are a favorite among children and adults alike. But how are these tasty candies made?
In this article, we explore the machines and processes that bring gummy bears to life in factories around the world.
1. Mixing Equipment
The journey of a gummy bear starts with the mixing of ingredients. A high-speed mixer or a planetary mixer is used to blend gelatin, sugar, water, flavorings, and colorings. These machines ensure that all ingredients are thoroughly combined to create a homogenous mixture.
2. Heating and Dissolving Units
Before the mixture can be poured into molds, the gelatin needs to be dissolved. This is done using a heating unit that gently warms the mixture to the right temperature, ensuring that the gelatin fully dissolves and the mixture is ready for the next step.
3. Depositors and Fillers
Once the mixture is ready, it’s time to fill the molds. Depositors or fillers are machines designed to precisely pour the gummy mixture into the molds. These machines can handle various shapes and sizes of molds, ensuring that each gummy bear is accurately formed.
4. Gummy Bear Molds
Molds are a crucial part of the process. They come in various shapes, including the iconic bear shape, and are often made from stainless steel for durability and easy cleaning. The molds are designed with individual cavities that will take the shape of each gummy bear.
5. Cooling Tunnels
After the molds are filled, they are sent through cooling tunnels. These tunnels rapidly cool the gummy mixture, allowing it to set and solidify into the desired shape. The cooling process is essential for achieving the chewy texture that gummy bears are known for.
6. Demolding Machines
Once the gummies have set, they need to be removed from the molds. Demolding machines carefully eject the gummy bears from their molds without damaging their shape. This step requires precision to ensure that each gummy bear maintains its integrity.
7. Quality Control Stations
Before the gummy bears move on to packaging, they pass through quality control stations. Here, machines and human inspectors check for any defects, such as misshapen gummies or inconsistencies in color and size. This step ensures that only the best gummy bears make it to the final packaging.
8. Coating and Enrobing Equipment
Some gummy bears are coated with a layer of sugar or chocolate. This is done using enrobing machines that dip the gummies into a chocolate or sugar coating, then spin them to remove excess coating and create a smooth, even layer.
9. Drying and Cooling
After being coated, gummy bears need to be dried and cooled again to set the coating. This is done in a separate drying and cooling area, which may use air circulation or other methods to gently dry the gummies without affecting their texture.
10. Packaging Machines
Finally, the gummy bears are ready for packaging. Automatic packaging machines wrap the gummies in individual wrappers or place them into bags, boxes, or other containers. These machines can also add labels, seals, and other packaging elements to ensure the gummies are ready for retail.
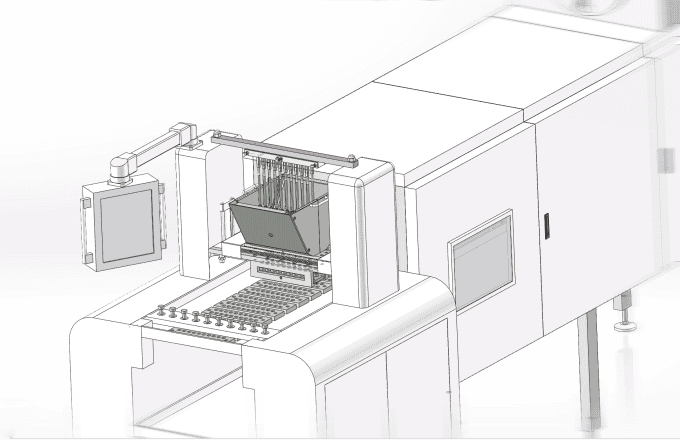
11. Metal Detectors and X-Ray Machines
For safety, gummy bears pass through metal detectors or X-ray machines to ensure there are no foreign objects or contaminants in the final product.
12. Conveyor Systems
Throughout the entire process, conveyor systems transport the gummy bears from one machine to another. These systems are designed to handle the weight and size of the gummies and molds, ensuring a smooth and efficient production line.
Conclusion
The production of gummy bears is a fascinating blend of art and science, with machines playing a crucial role in every step of the process. From mixing and molding to cooling and packaging, each machine is designed to ensure that gummy bears are produced efficiently, safely, and to a high standard of quality. The next time you enjoy a gummy bear, you can appreciate the sophisticated machinery and processes that brought it to life.